 This tells us that there must be something else in the nucleus as well as protons.įor small atomic numbers, the relative atomic mass is approximately double the atomic number. However, the atomic mass is not proportional to the atomic number (see page 6). Their atomic numbers correspond to their position in the sequence (as well as to the number of protons in their nuclei). The elements in the periodic table are arranged in increasing atomic mass. The atomic number is written to the left bottom of the symbol. (The symbol for lithium is Li - note it starts with a capital letter although its name, lithium, starts with a small letter.) We can show this information about an atom using its symbol and atomic number: Any atom that has 3 protons must be a lithium atom. Therefore, the atomic number of lithium is 3. This number is the atomic number of the element and we give it the symbol Z.įor example, lithium atoms contain 3 protons. Some atoms and their atomic numbers.Īll atoms of a given element have the same number of protons in the nucleus. Picture 1.7 Lithium has 3 positve protons in its nucleus. The neutrons help keep the nucleus together. The neutron has a similar mass to the proton but has no charge. For example, if a nucleus contains three protons, then it is part of an atom of lithium (atomic number 3). It corresponds to the atomic number of that element. The number of protons in a nucleus is the same for all the atoms of a particular element. The proton has a positive charge and a large mass (1800 times more than an electron see table 1). There are two types of particle in the nucleus of an atom: the proton and the neutron. The relative mass(m) and the relative charge (q) of the three main subatomic particles. 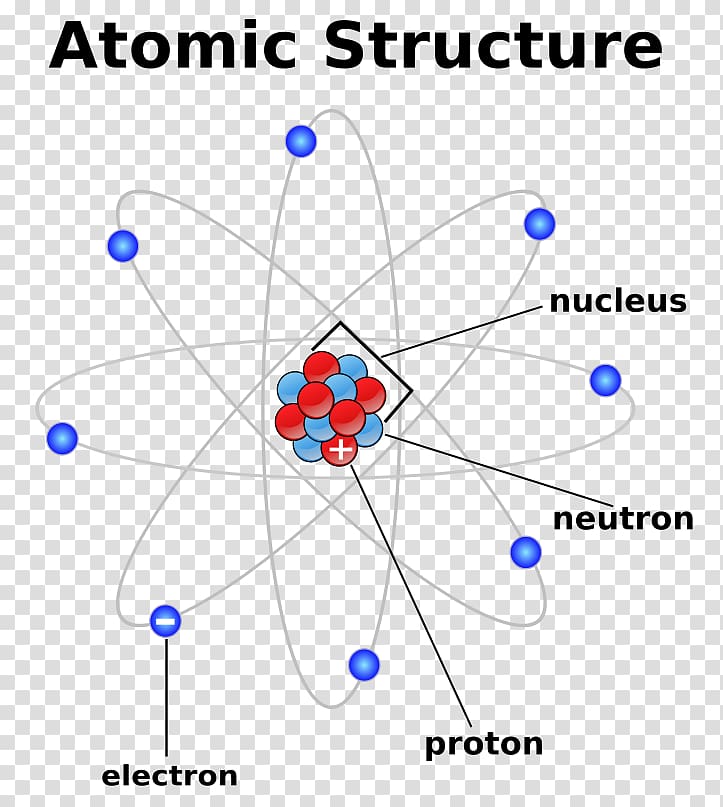
The positive charge in the nucleus is balanced by the negative electrons, making an atom neutral.īut what's inside the nucleus? And what are the fundamental particles that make up an atom? Most of the mass of the atom is concentrated in the nucleus, which has a positive charge. The electrons have a negative charge and very little mass. The atom is made from a central nucleus with electrons around the outside. Picture 1.6 An atom has a nucleus with electrons outside it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
